













Acetic acid, systematically named ethanoic acid, is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH. Vinegar is at least 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water. It has been used, as a component of vinegar, throughout history from at least the third century BC. Acetic acid is also known as acetyl hydroxide (AcOH).

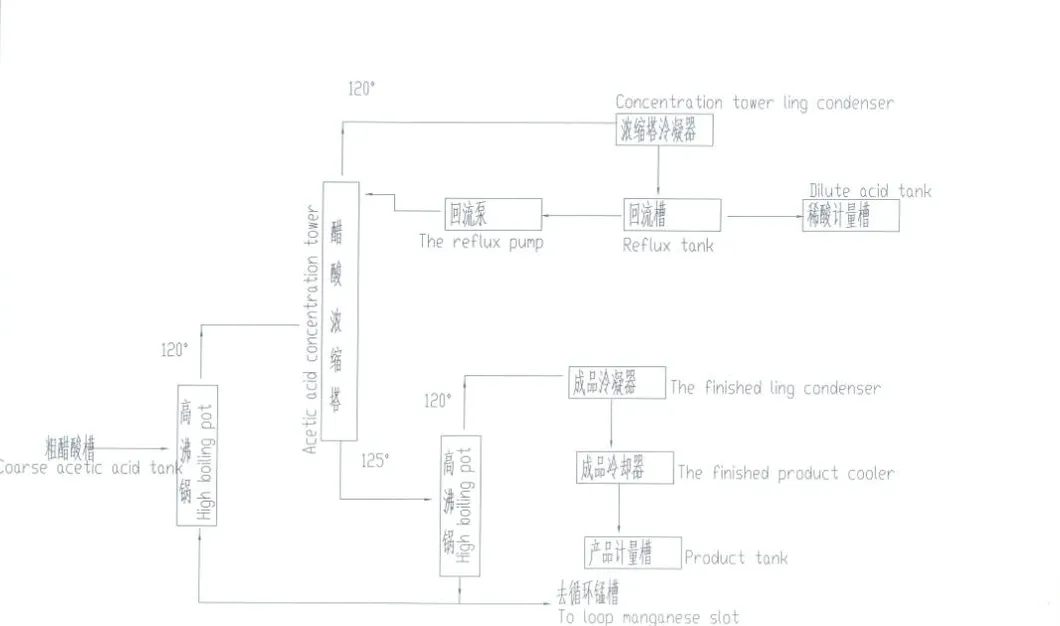
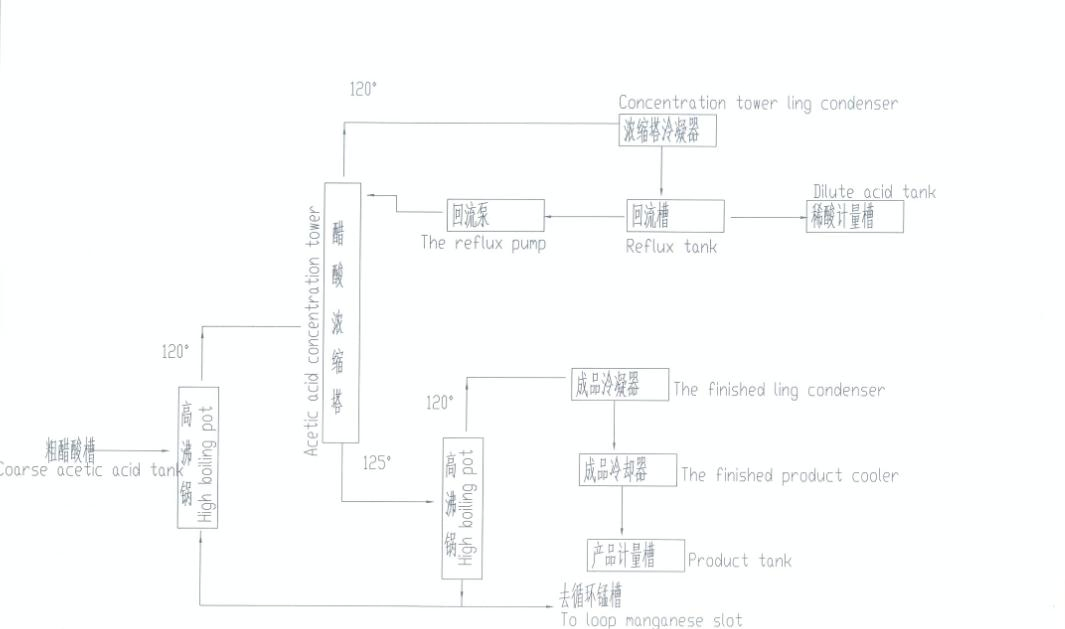
Acetic acid is produced industrially both synthetically and by bacterial fermentation. Approximately 75% of industrial acetic acid is produced via methanol carbonylation.
This process involves methanol and carbon monoxide reacting to produce acetic acid. Modern plants utilize the Cativa process (iridium-catalyzed) which is greener and more efficient than the older Monsanto process.
Chemical Equation: CH3OH + CO → CH3COOH
Though less common than carbonylation, this remains a significant manufacturing method where acetaldehyde is oxidized using metal catalysts like manganese or cobalt salts.
A single-stage conversion of ethylene to acetic acid using a palladium metal catalyst. This method is highly competitive for smaller production plants.